Without electrolysis there is no green hydrogen and thus no nationwide and long-term reduction of emissions - but how is hydrogen actually produced? What is an electrolyzer and what types are there? What role do cell voltage and current density play in electrolysis and what contribution does DiLiCo engineering's measurement technology make to improving this technology? These questions will be answered in the following.
What are electrolyzers?
The electrolyzer is one of the essential hydrogen technologies of a hydrogen economy. With its help, water can be split into its components hydrogen and oxygen. Electrolysis thus represents the reverse reaction of the fuel cell. An electrolyser consists of several electrolysis cells arranged in a row, in which the necessary redox reaction is forced by supplied electrical energy.
The working principle of electrolysis was already discovered at the beginning of the 19th century. The potential of hydrogen as an energy carrier was already recognised at that time. The French writer Jules Verne recognised the potential of water and hydrogen as early as 1875 in his novel The Mysterious Island:
"Water is the coal of the future. Tomorrow's energy is water that has been decomposed by electric current. The elements of water thus decomposed, hydrogen and oxygen, will secure the earth's energy supply for the unforeseeable future." (Jules Verne, 1875)
Upcoming events with DiLiCo
How are electrolyzers constructed?
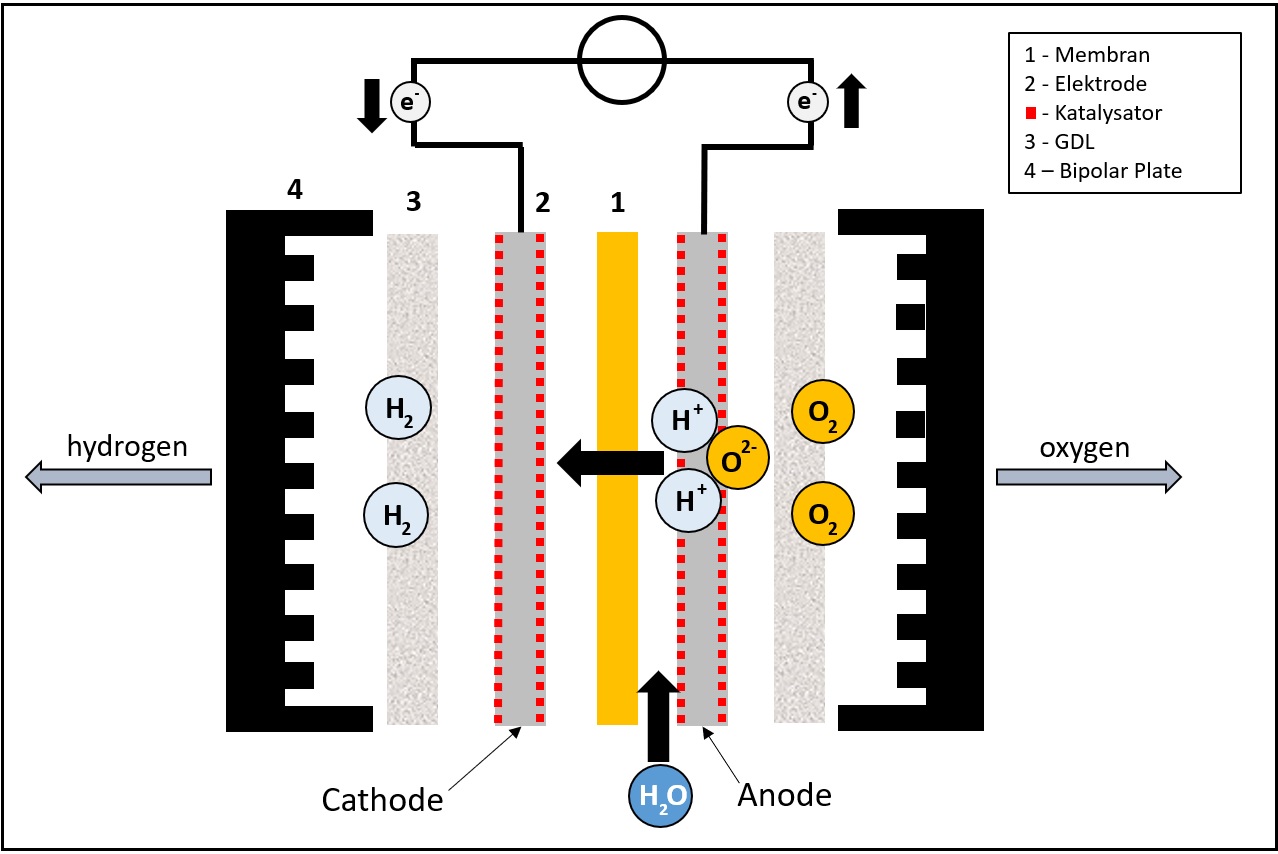
The structure of an electrolysis cell is similar to the structure of fuel cells. In PEM electrolysis, the electrolysis cell consists of two electrodes, the anode and the cathode, which are spatially separated from each other by a partially permeable membrane. To accelerate the speed of reaction, there are catalysts on both sides of the membrane. Only few noble metals such as platinum and iridium can be used here, as other metals would convert too quickly under the harsh reaction conditions. The outer envelope of the electrolytic cell is formed by the bipolar plates, which must also be made of corrosion-resistant material such as stainless steel or titanium.
How electrolysis works?
An external voltage is applied to the electrodes of the electrolysis cell, which must be at least above the decomposition voltage of water (usually 1.6 V - 2 V). Accelerated by the catalytic effect of the precious metal electrodes, water supplied to the anode is split. Oxygen, free electrons and positively charged hydrogen ions are produced.
The positive hydrogen ions then migrate through the membrane to the cathode, where they are reduced to molecular hydrogen. The hydrogen can then be taken from the cell and stored. You can find out what options there are for hydrogen storage in our article on hydrogen technologies.
What variants of electrolyzers are there?
In electrolysis, there are various processes for producing hydrogen. These differ mainly by the electrolyte used, the operating temperature and by the structure of the electrolysis cell. The three best-known types of electrolysis are described below and their mode of operation briefly explained.
AEL - Alkaline electrolysis
The Alkaline ELectrolysis (short: AEL) uses as electrolyte a potassium hydroxide solution and a permeable membrane, the so-called diaphragm, which separates the two electrodes from each other. The electrodes are immersed in an alkaline aqueous solution. If a voltage is applied, oxygen is produced at the anode and hydrogen at the cathode.
The great advantage of this form of electrolysis is that the technology has high long-term stability and low investment costs. The efficiency of alkaline electrolysis is currently around 65% and up to 90,000 operating hours can be achieved. The electrolysis plants can be in the power range of up to 130 megawatts and are already in use worldwide.
PEM - Proton-Exchange-Membran electrolysis
The proton exchange membrane electrolysis (Proton-Exchange-Membrane, abbreviated: PEM) or also called polymer electrolyte membrane electrolysis uses a thin membrane made of thermoplastic (ionomer) as electrolyte. Anode and cathode are separated by this gas-tight membrane, only positive hydrogen ions can pass through the membrane. Due to ion migration, this process of electrolysis belongs to the acidic processes. This makes the use of precious metals necessary for the catalysts in order to avoid corrosion.
The advantage of this technology is the good load change behaviour. It can react quickly to fluctuations in the energy supplied and can be operated at partial load without any problems. The efficiency of PEM electrolysis is currently around 63%, which is slightly lower than that of alkaline electrolysis. Since the technology is still relatively new, the investment costs are considerably higher than for alkaline electrolysis. The power range of the plants reaches values of up to 6 megawatts.
SOE - Solid Oxide electrolysis
Solid oxide electrolysis (Solid Oxide Electrolysis, abbreviated: SOE) uses a solid ceramic material as the electrolyte, which separates the two half cells. The water is fed to the reaction chambers in the form of steam. This type of electrolysis is currently in transition from research to industrial application.
This technology belongs to the high-temperature electrolysis, is operated at temperatures of 600-900 °C and can achieve a very high efficiency of over 80%. The investment costs are about the same as for PEM electrolysis. Recently, the largest SOE electrolyser in the world with a power of 250 kW was commissioned by the company Sunfire GmbH.
Applications of electrolyzers for hydrogen production
The applications of electrolysers take place where hydrogen production makes sense from a logistical and energetic point of view. For example, at large photovoltaic plants or at wind farms. There, the regeneratively generated electricity can be used for on-site water electrolysis. The hydrogen produced is usually stored under high pressure so that it can be transported on to the user. End users can be, for example, the mobility sector or also steel production in order to reduce CO2 emissions.
Another possible application of electrolysers is the production of hydrogen, which is fed into the natural gas grid. Up to 10 % hydrogen can be fed directly into the natural gas grid. Via the methanisation step, hydrogen can also be converted into pure natural gas beforehand. Furthermore, electrolysis is used in the chemical industry to produce gases for production processes.
Electrolyzers belong to the so-called Power-to-Gas (short: PtG) (or also Power-to-X) technologies, in which electricity is converted into different gases (or different forms of energy) and stored. In the case of electrolysis, electricity is converted into the form of hydrogen.
What role can electrolyzers play in the energy transition?
The PtG technology can be a key technology for the energy transition. Through it, different sectors can be coupled to save emissions at several points like the gas, heat and mobility sectors. On the part of policy, the change towards hydrogen technologies and a hydrogen economy is required. However, it depends on how the hydrogen is produced and where it is produced. Most of the time, hydrogen is not needed where it can be produced. It must therefore be stored and transported.
All these energy expenditures must be included in the effect chain. Therefore, it must be carefully considered at which points the production and application of hydrogen technologies make sense. The fact that the technology of electrolysis represents an important form of energy storage can no longer be denied. In addition to the recovery of electrical energy through fuel cells, hydrogen can also be integrated into various other sectors on a transitional basis.
What are the research priorities and the biggest challenges?
Current research is concerned with the question of optimal scaling of the technology as well as with the processes of series production of the components, some of which are very difficult to produce. In addition, improving the lifetime of the systems is always an important research focus. The development of new forms of electrolysers is also being pushed forward in research. The biggest challenges of the technology are to get it ready for mass production and to scale it up further to bring down the costs. Only then will it be possible to produce green hydrogen at a sufficiently cheap level. Only then can electrolysis and green hydrogen become competitive.

Image: The measurement device DiLiCo current density for the insight into the cell.
The role of current density in electrolysis
The current density within a PEM electrolysis cell plays an important role in the safe and efficient operation of the electrolyser. This is because the current density correlates with the overvoltage of the cell. If this is too low, electrolysis cannot take place and the water is not split.
The highly specialised measuring system DiLiCo current density gives the opportunity to take a look inside the cell. It shows how the current density is distributed over the reaction surface and thus allows conclusions to be drawn about the ageing of the cell as well as optimal operation. This has decisive advantages for the user:
- Analysis of the reaction within the cell
- Optimisation of the mode of operation of the electrolysis
- Insight into current density and temperature behaviour within the cell
DiLiCo engineering adapts the current density measurement system to any requirement. DiLiCo engineering aims to design a perfectly adapted measurement system for each customer, which provides valuable insights into their electrolysis cells.
The role of cell voltage in electrolysis
The cell voltage of the individual electrolysis cells in an electrolyser plays a very important role. The cells must maintain a certain cell voltage, the so-called decomposition voltage, at which the water is split into oxygen and hydrogen. Maintaining the decomposition voltage is essential for the smooth operation of the electrolyser. The increasing ageing of cells can also be seen in the level of the cell voltage and must be permanently controlled for smooth operation.
The measuring device DiLiCo cell voltage fulfils exactly this requirement. The application of single cell voltage measurement has the following advantages for the user:
- Safe operation of the electrolysis by maintaining the decomposition voltage
- Regulation of the cell voltage through communication with the controller
- Avoidance of undesired downtime and consequential costs of the plant
Image: The measurement device DiLiCo cell voltage for single cell voltage measurement.

 Deutsch
Deutsch